From delays to delivery: Accelerating study activation for clinical research sites
For academic medical centers (AMCs) and health systems, clinical research is a critical pillar for institutional reputation and financial health. However, while the science of medicine is advancing quickly, the administrative process required to start a trial is struggling to keep up.
The average site initiation time for Phase 1-3 trials at AMCs and hospitals stretches to a lengthy 9.4 months.1 This stands in stark contrast to ambitious guidelines, such as the National Cancer Institute's 90-day target. This six-month activation gap represents more than just lost time; it represents lost revenue, lost sponsor trust, and, most importantly, lost opportunities for patients awaiting life-saving therapies.
Many site leaders attempt to solve startup delays by adding more coordinators or pushing teams to simply work faster. However, the bottleneck is rarely due to a lack of effort, but rather, from the compounding complexity of the modern clinical trial.
Today’s activation process involves a number of stakeholders:
- Regulatory and Investigational Review Board (IRB): Navigating increasingly complex compliance landscapes.
- Legal and finance: Managing rigorous budget negotiations and coverage analysis.
- Ancillary services: Coordinating with pharmacy, labs, and imaging — all of whom have their own internal queues.
- Principal Investigator: Balancing clinical duties with the administrative burden of trial oversight.
When these departments operate in silos, information is passed like a baton in a relay race — often dropped, frequently delayed, and usually invisible to the leadership team until a deadline is missed. Coordinating between disparate spreadsheets, email threads, and legacy clinical trial management systems (CTMS) can lead to prolonged activation times.
The hidden costs of delayed study activation
The impact of the activation gap extends far beyond the research office. To understand the true value of shortening this cycle, leadership must look at three specific areas of risk:
1. Financial impacts
Every month a trial sits in the startup phase is a month of zero revenue for a study that has already incurred costs in the form of administrative labor. For sites operating on thin margins, the inability to move quickly from a signed contract to a billable patient visit can lead to significant budgetary shortfalls.
2. Reputational risks
A research site’s ability to rapidly activate studies is an important selection factor for sponsors and CROs. After all, every day a clinical trial is delayed can cost sponsors an estimated $800,000 in lost revenue opportunities.2 If a research site consistently takes 200+ days to activate, they’re likely being deselected for high-profile trials before they even receive the feasibility questionnaire.
3. The patient access gap
For patients with late-stage or rare diseases, time is of the essence. When a trial activation is delayed by six months due to administrative friction, it can limit access to potential cures, making operational efficiency critical to patient care.
Shifting from reactive to proactive management
To close the activation gap, sites must move away from status update culture where teams meet weekly to discuss what happened in the past. The evolution should be toward a predictive culture, where teams use data to proactively manage study activation workflows.
True innovation in study startup involves:
- Standardization: Identifying the specific milestones (e.g., budget approval, IRB submission) that actually drive the timeline, rather than getting lost in administrative noise.
- Centralized visibility: Creating a single source of truth where all teams can see bottlenecks during study activation.
- Proactive management: Using historical data to identify common challenges during study activation to refine strategic workflows that can lead to faster study activation in the future.
The faster path forward — less time, same quality
The 9.4-month average is not a law of nature; it’s a symptom of fragmented processes and outdated tools. As clinical trials grow more complex — spanning decentralized models, real-world data, and advanced biomarkers — sites that succeed will be those that make activation speed a core capability, not a bottleneck.
Solving the complexity of study startup requires a blueprint for operational excellence.
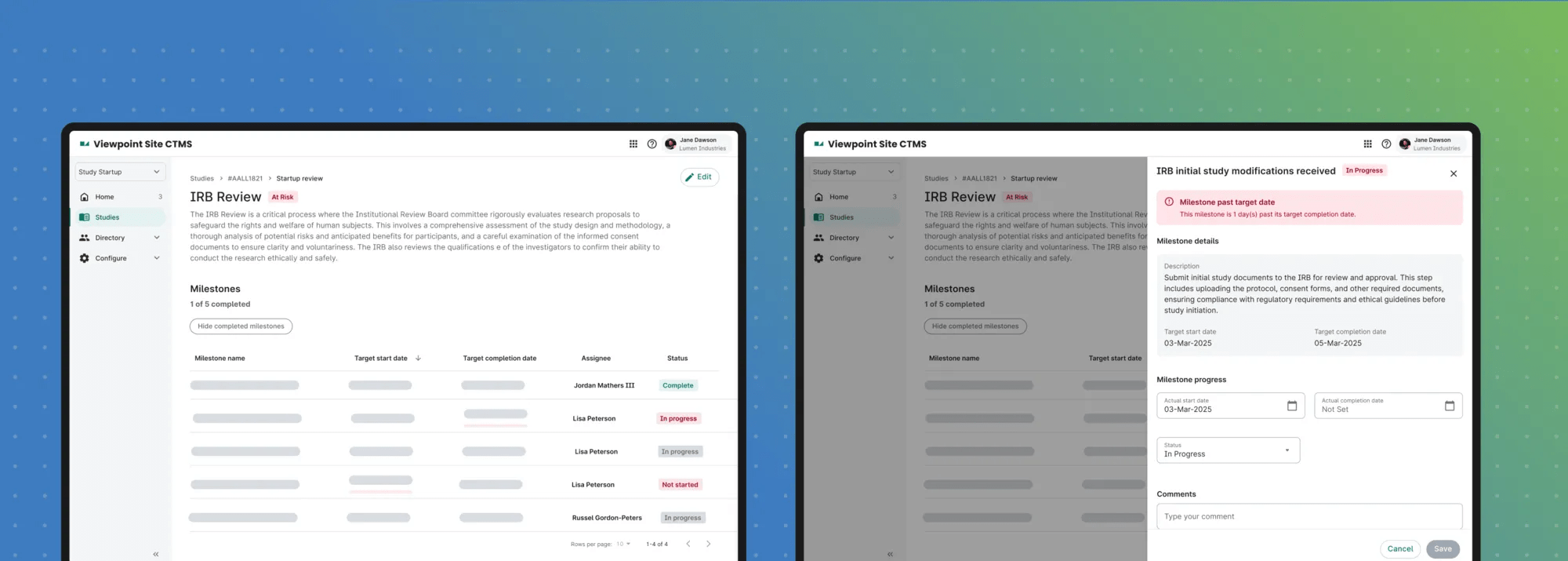
Solutions like Verily Viewpoint Study Start Up for Sites are designed to address these industry challenges head-on, bringing structure to complexity, connecting workflows, and helping sites move faster without sacrificing rigor. Drawing from this approach, Verily has identified the key drivers of activation delays and outlined a practical roadmap for sites targeting the 90-day NCI benchmark.
Download our whitepaper, Built for sites: Accelerated study activation to revolutionize trials, to explore:
- How to build a customizable critical path for complex therapeutic areas.
- Ways of integrating activation workflows to eliminate manual tracking.
- Strategies for meeting PRMS requirements without sacrificing speed.
Sources
1 ACRP. “Overcoming Study Start-Up Delays: Best Practices for Research Sites.” ACRPnet.org, May 7, 2025.
https://acrpnet.org/2025/05/07/overcoming-study-start-up-delays-best-practices-for-research-sites
2 Smith, Zachary, Joseph A. DiMasi and Kenneth Getz. Quantifying the Value of a Day of Delay in Drug Development. Tufts Center for the Study of Drug Development, August 2024.
https://csdd.tufts.edu/sites/default/files/2025-02/Aug2024%20Day%20of%20Delay%20White%20Paper%20Final.pdf?1744237947=


